The EHEC-host interactome reveals novel targets for the translocated intimin receptor
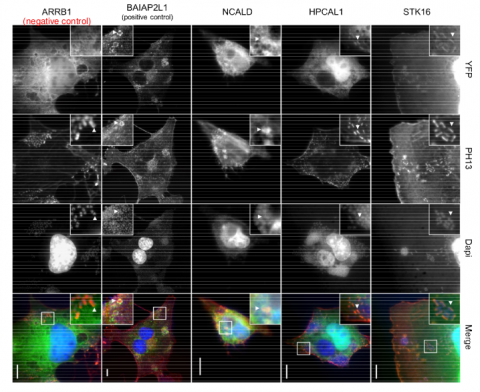
Blasche S, Arens S, Ceol A, Siszler G, Schmidt MA, Häuser R, Schwarz F, Wuchty S, Aloy P, Uetz P, Stradal T, Kögl M, Enterohemorrhagic E. coli (EHEC) manipulate its human host through at least 39 effector proteins. Since these effectors hijack host processes through direct protein-protein interactions we performed comprehensive yeast two-hybrid screens to investigate 34 effectors, allowing us to find 48 high-confidence protein-protein interactions between 15 EHEC effectors and 47 human host proteins. In comparison to other bacteria and viruses we found that EHEC effectors bind more frequently to hub proteins as well as to proteins that participated in a higher number of protein complexes. While five interactions were previously reported we also found novel interactions of EspJ and EspY as well as six new interactions that involve the translocated intimin receptor (TIR), namely HPCAL1, HPCAL4, NCAL4, ARRB1, PDE6D, and STK16. We also compared these TIR interactions in EHEC and enteropathogenic E. coli (EPEC) and found that five interactions were conserved. Notably, the conserved interactions included those of serine/threonine kinase 16 (STK16), hippocalcin-like 1 (HPCAL1) as well as neurocalcin-delta (NCALD), proteins that we also found to co-localize with the infection sites of EPEC. Furthermore, our results suggest putative functions of poorly characterized effectors (EspJ, EspY1). In particular, we observed that EspJ is connected to the microtubule system while EspY1 appears to be involved in apoptosis/cell cycle regulation.
Scientific Reports,
2014, 4: 7531
Pubmed: 25519916
Direct link: 10.1038/srep07531
